Please wait while flipbook is loading. For more related info, FAQs and issues please refer to DearFlip WordPress Flipbook Plugin Help documentation.
Introduction
Nuclear Energy refers to the energy stored in the Nucleus of atoms and a Nuclear Reaction refers to a process by which such energy is released, as the result of the transformation from one type of substance to another The best examples of Nuclear Reactions is found in nature within Stars, such as the Sun of our Solar System. The Sun act as a natural Nuclear Reactor, whereby energy can be released as a result of fusing atoms together and/or splitting individual atoms apart.
Before we continue, it is important to explain what an Atom is. An Atom is the basic building block of matter that cannot be seen with the human eye. Atoms can be likened to a child’s wooden or plastic toy bricks, or to concrete bricks used as the basic unit of a structure such as a house. Atoms are so small that you will find millions of them on a pin head. It is important to note that the atoms can be broken down into even smaller units such as Protons, Neutrons and Electrons. An atom is composed of a central Nucleus containing protons and neutrons. Orbiting around the nucleus are electrons. The proton is a positively charge particle and the electron is a
negatively charged particle. The neutron is said to have no electrical charge. As mentioned, Protons and Neutrons exist within the Nucleus and are known as Nucleons. The Nucleons themselves can also be divided into even smaller particles, known as Quarks. The Nucleons (Protons and Neutrons) are held together in the Nucleus by the Strong Nuclear Force, which is one of the Four Fundamental Forces. (Strong Force, Weak Force, Electric Attraction (Electromagnetism) and Gravity).
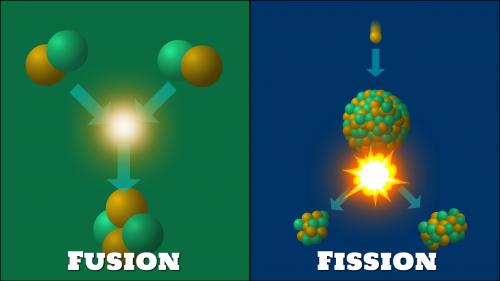
Nuclear Fusion is a reaction where two or more atomic nuclei combine or fuse to form heavier nuclei and releases energy as a result of the process. This occurs in nature in stars, such as the sun within our own solar system.
The sun fuses hydrogen atoms together in order to produce helium. The high temperature (Approximately 100 million Kelvin) is responsible for a form of matter known as plasma, which is a gaseous substance where electrons exist separately from hydrogen atomic nuclei. As the atomic nuclei exist primarily as protons, which are positively charged, their nature is to repel each other. This is similar to attempting to bring two like poles of a magnet together. The sun will have stronger magnetic forces that will force the atomic nuclei to fuse together producing helium. This is the beginning of synthesizing the chemical elements as defined in the Periodic Table of Elements
Nuclear Fission is a reaction, where the heavy nucleus of materials such as Uranium or Plutonium breaks into smaller parts as a result of a collision with subatomic particle and energy is released. A typical nuclear reactor will work by projecting neutrons at high speed to collide with atoms. Once the collision has taken place, an atom will split, releasing its own neutron/s to collide with other atoms and so on. This causes what is known as a Chain Reaction, which will continue as long as the required atoms are available. The reactor will have a system that can control the chain reaction using control rods. These rods are able to absorb neutrons and are typically made from elements such as Boron and Cadmium. Water can also be used to control a nuclear reaction by cooling the temperature. An uncontrolled nuclear reaction is equivalent to the explosion of an atomic bomb. The energy released is thermal and is 100 million times more powerful than a normal chemical reaction.
Radioactive Decay is a natural spontaneous occurrence when unstable atomic elements or their Isotopes break down into different Nuclei while emitting energy. This process is described as spontaneous because modern day scientist are unable to accurately predict when decay will begin naturally. However, the amount of time that materials remain Radioactive can be estimated by calculating the Half Life. Also, it is important to note that the released radioactive energy is Ionising Radiation, meaning that certain levels of exposure can be extremely harmful to humans.
There are three main types of radioactive decay: Alpha, Beta and Gamma
Alpha Decay is where unstable atomic nuclei, eject what is equivalent to Helium nuclei having no electrons, which is composed of a combination of 2 protons and 2 neutrons, and having done this, transforms into a different stable element. The ejected energy is referred to as an Alpha Particle. For example, an unstable Uranium atom (atomic number 92) will decay by emitting an alpha particle and transforming into Thorium (atomic number 90). The penetration levels of alpha particles are such that they can be blocked by a few sheets of paper. Alpha particles can be used in devices such as smoke detectors, nuclear batteries to generate electricity and devices that remove Static Electricity from tools.
Beta Decay is where unstable nuclei release energy by ejecting Electrons as a result of a transformation of Neutrons into a Protons or vice versa. This introduction of an additional proton into the nucleus will cause a transformation into the next chemical element for an increasing atomic number, whereas the introduction of an additional neutron will cause a transformation into the previous chemical element for a decreasing atomic number. For example an unstable Strontium atom (atomic number 38) will decay by emitting a beta particle and transforming to Yttrium (atomic number 39). The penetration levels of beta particles are such that they can pass
through paper but will be blocked by Aluminium foil. Beta particles can be used to measure the thickness of materials and can also be used as Tracers within the medical industry.
Gamma Decay is another type of radioactive decay that an atomic nucleus can undergo. What separates this type of decay process from alpha or beta decay is that no particles are ejected from the nucleus when it undergoes Gamma Decay. Instead, a high energy form of electromagnetic radiation, a gamma ray photon, is released. The penetration levels of gamma particles are such that they can only be blocked by materials such as thick concrete or Lead. Uses for gamma energy are found in industrial sterilisation and in nuclear power.
The three main areas where the dangers of nuclear energy need to be thought about are as follows: Nuclear Reactors, Atomic Bombs, Hadron Colliders
Nuclear Reactors are mostly used as Fission Reactors in Nuclear Power Stations to generate electricity. These power stations house unstable Radioactive material, such as Uranium and Plutonium. Nuclear Reactors are also used in research or in Nucleosynthesis, which is a process where alternate Elements are produced from pre-existing elements. The concerns here include the fact that there is always a risk of an unforeseen incident occurring, causing a disaster such as a Meltdown, where radiation escapes into the surrounding atmosphere. These not only cause sickness and death, but also cause the surrounding area to be uninhabitable for decades to come. The following is a list of some of these incidents; 1957 Kyshtym (Russia), 1957 Windscale (UK), 1986 Chernobyl (USSR), 1979 Three Mile Island (UK), 2005 Artucha (Argentina), 2005 Sellafield (UK), 2011 Fukushima (Japan).
Atomic Bombs are Weapons of Mass Destruction (WMDs), that when used cause Nuclear Explosions resulting in uncontrolled “Chain Reactions”. The history of the Atomic Bomb does not make good reading, where once scientist discovered how to split the Atom, some bright spark decided to drop Atomic Bombs on the Japanese cities of Hiroshima and Nagasaki. These incidents, in 1945, where the intense heat initially killed hundreds of thousands and the subsequent radiation caused sickness, such as deformities and genetic defects to the Japanese inhabitants for generations to come.
Hardron Colliders are Particle Accelerators, which are machines that fire sub-atomic particles at increasing high speed in order to smash into other sub-atomic particles. This is said to be done for experimental purposes in an attempt to recreate events such as “The Big Bang”. Hadrons are a sub-atomic particles such as Protons and Neutrons, which themselves are composed of Quarks. Currently, the most well know Hadron Collider is the LHC (Large Hadron Collider) CERN, is a 27 kilometre circular super machine which exist underground on the border between France and Switzerland near Geneva.
The problem here is that, no matter how many tests or simulations are done, the resulting outcome of particle smashing is extremely unpredictable, where modern scientists are still “babies” when it comes to artificially creating Black Holes, Dark Energy, Dark Matter and Quantum Physics



